Learn about how Dr. Shvartsburg and GAA Custom Engineering are making it possible to achieve high-resolution FAIMS.
High-Resolution FAIMS Pre-Filter for Mass Spectrometers
Ion mobility spectrometry (IMS) separates isomers and isobars prior to MS analyses.
Differential or Field Asymmetric waveform IMS (FAIMS) that employs high electric fields is much more orthogonal to MS than conventional (drift tube or traveling-wave) IMS using low fields and thus allows better separations at same formal resolving power. However, that advantage had been offset by lower FAIMS resolving power.
Over the last decade, Alex Shvartsburg and Gordon Anderson worked to develop high-resolution planar-gap FAIMS, combining new mechanical designs, electronic circuitry, and operational regimes to raise the resolving power up to ~500 [1]. This performance has enabled previously unthinkable analyses, such as baseline separation of PTM localization isomers for modified peptides [2], individual protein conformers [3], lipid regioisomers [4], and even isotopomers [5]. The capability for such fine separations is poised to open new avenues in chemical and biological analyses.
GAA Custom Engineering, in collaboration with Dr. Shvartsburg, is now making high-resolution FAIMS (with ESI ion source) available for research in proteomics, metabolomics, and other areas. The new systems, comprising drivers protected against electrical breakdown, allow unconstrained exploration of carrier gas compositions without damaging discharges. The stages, operating at ambient pressure and temperature, can be coupled to many mass spectrometers with minimum front-end modifications. GAACE will work with customers to define and implement the needed modifications and integrate the FAIMS systems with existing mass spectrometers.
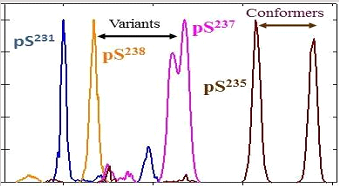
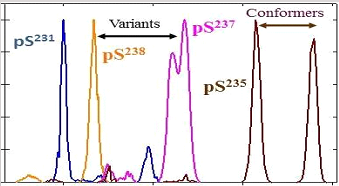
Separation of all four PTM localization variants for a tryptic phosphopeptide from human τ protein
A. A. Shvartsburg et al., Anal. Chem. 83, 5078 (2011)

Complete FAIMS/ LTQ XL system with ETD capability
1. A. A. Shvartsburg et al. J. Am. Soc. Mass Spectrom. 24, 109 (2013).
2. A. A. Shvartsburg, Y. Zheng, R. D. Smith, N. Kelleher. Anal. Chem. 84, 4271 (2012).
3. A. A. Shvartsburg. Anal. Chem. 86, 10608 (2014).
4. A. A. Shvartsburg et al. J. Am. Soc. Mass Spectrom. 22, 1146 (2011).
5. A. A. Shvartsburg, D. E. Clemmer, R. D. Smith. Anal. Chem. 82, 8047 (2010).
For inquiries, contact gaa@owt.com.